The Fundamental Difference: Biosimilars vs Generics
To understand why making a biosimilar is so hard, we have to look at the difference between a small-molecule drug and a biologic. Most generics are small molecules. Think of them like a bicycle-simple, predictable, and easy to build from a blueprint. If you follow the chemical formula, you get the exact same molecule every time. Biosimilars are biologic medical products made from living organisms, such as bacteria or yeast, which result in large, intricate protein structures. If a generic is a bicycle, a biosimilar is a Boeing 747. You can't just "copy" a living cell. These molecules are massive and fold into complex 3D shapes. Because they come from living systems, there is inherent variability. This means manufacturers can't just use a chemical synthesis; they have to manage a biological environment where the cells are essentially the factory workers. If the "workers" are stressed or the temperature shifts by a degree, the final protein might fold incorrectly, rendering the batch useless.The Glycosylation Puzzle: A Tiny Detail with Big Risks
One of the most frustrating technical hurdles is Glycosylation, which is the process where carbohydrate structures, or glycans, attach to the protein backbone of a biologic. These sugar chains are not just decorative; they act like a GPS for the drug, telling it where to go in the body and how long to stay there. Glycosylation is incredibly sensitive. A manufacturer has to balance a dozen variables simultaneously:- The specific type of host cell used for production.
- The exact nutrient mix in the culture media.
- Dissolved oxygen levels and pH balance.
- Consistent temperature control throughout the growth cycle.
The Scale-Up Trap: From Lab to Factory
Moving a process from a small 2-liter laboratory flask to a 20,000-liter Bioreactor, which is a stainless steel or single-use vessel designed to support mammalian or microbial cell growth, is where many projects fail. This is known as the scale-up challenge. In a small flask, oxygen and nutrients reach every cell almost instantly. In a massive commercial tank, the physics change. You get "dead zones" where the liquid doesn't mix well, or "hot spots" where the temperature varies. The cells at the bottom of the tank experience different pressure and oxygen levels than the cells at the top. To fix this, engineers have to meticulously adjust stirring speeds and aeration rates so the cells "feel" the same environment regardless of the tank size. Furthermore, not every factory has the space or the budget for these massive installations. For mid-sized companies, investing in a new production hall just to accommodate a larger bioreactor can be a financial gamble that requires millions in upfront capital.| Feature | Generic Drugs | Biosimilars |
|---|---|---|
| Structure | Simple, small molecules | Large, complex proteins |
| Production | Chemical synthesis | Living cell cultures |
| Consistency | Identical every time | Highly similar, but variable |
| Development | Relatively fast and cheap | Long, expensive, and technical |
| Sensitivity | Stable to most conditions | Extremely sensitive to temp/pH |
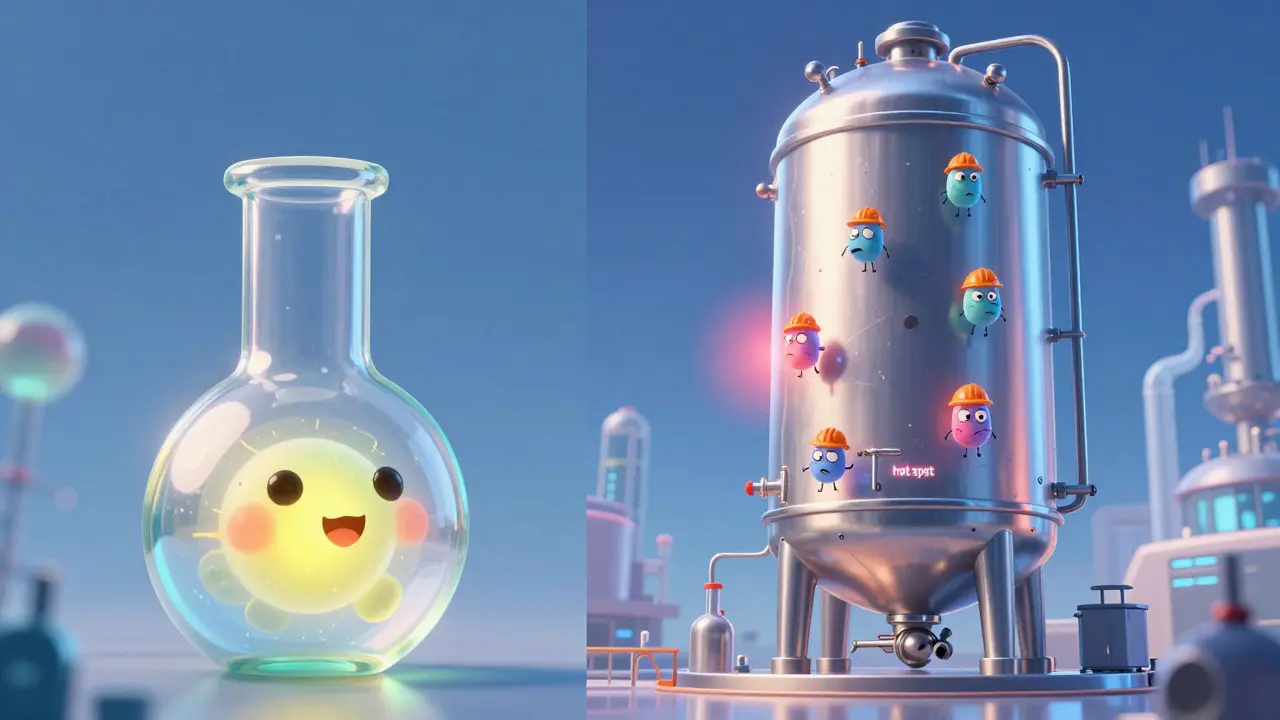
Cold Chain and Batch Consistency
Once the protein is grown, the nightmare isn't over. Biologics are fragile. They are sensitive to light, shaking, and-most importantly-temperature. This necessitates a strict Cold Chain, which is a temperature-controlled supply chain used to maintain the integrity of a product from manufacture to delivery. If a shipment of biosimilars sits on a hot loading dock for two hours, the proteins can denature (unfold), making the drug ineffective or dangerous. The risk of damage is highest during the filling and transport stages. A single bag breakage in a bioprocessing container can result in the loss of a batch worth millions of dollars. Achieving batch-to-batch consistency is the final boss of manufacturing. In chemical synthesis, if you put in X and Y, you get Z. In biological production, the cells are living entities. They can drift over time. This means manufacturers must implement Process Analytical Technology (PAT), using real-time sensors to monitor the cells and tweak the environment on the fly to keep the output consistent.Navigating the Regulatory Maze
Getting a biosimilar to market isn't just about the science; it's about the paperwork. Regulatory agencies like the FDA require a massive amount of evidence to prove that the biosimilar is "highly similar" to the reference product. This involves a three-pronged approach: analytical studies, preclinical tests, and clinical trials. Manufacturers must use advanced analytical techniques to prove that the critical quality attributes (CQAs)-like the aforementioned glycosylation and folding-are within a specific range. Because regulations change and differ between countries (e.g., FDA in the US vs. EMA in Europe), companies often have to run different sets of tests for different markets. This adds a layer of administrative complexity that can delay a launch by years.
Modern Solutions: Automation and Single-Use Tech
To survive these challenges, the industry is moving away from permanent stainless-steel tanks and toward Single-Use Technologies, which are disposable plastic bioreactors and bags used to prevent cross-contamination and speed up production changeovers. Single-use tech is a game-changer for a few reasons:- No Cleaning Validation: You don't have to spend weeks proving a tank is sterile; you just throw the plastic liner away and put in a new one.
- Faster Turnaround: Companies can switch from producing one biosimilar to another in a fraction of the time.
- Reduced Risk: Closed automated systems minimize human touch, which is the primary source of contamination in a cleanroom.
Why aren't biosimilars exactly the same as the original drug?
Because they are made in living cells, they have an inherent biological variability. Even the original "reference" product varies slightly from batch to batch. As long as the differences don't affect safety or how the drug works, it is considered biosimilar.
What is the biggest risk in biosimilar production?
The biggest risk is a failure in glycosylation or protein folding. If the molecular structure is wrong, the patient's immune system might attack the drug, or the drug might simply fail to treat the condition, potentially leading to a relapse of the disease.
How does scale-up affect the quality of a biosimilar?
Moving to larger tanks changes the physics of the environment. Issues like poor oxygen distribution or temperature fluctuations in a 20,000L tank can stress the cells, which in turn changes the protein quality compared to a small lab sample.
What is the role of the cold chain in biosimilars?
Biosimilars are proteins, and proteins denature (break down) when exposed to heat or extreme shaking. The cold chain ensures the drug stays at a precise temperature from the factory all the way to the pharmacy to prevent the medication from becoming inactive.
Why are single-use bioreactors preferred over stainless steel?
They eliminate the need for costly and time-consuming cleaning and sterilization validation. They also significantly reduce the risk of cross-contamination between different product batches, making the factory more flexible.


Divine Manna
April 5, 2026 AT 07:42The analogy of the Boeing 747 is quaint, yet it barely scratches the surface of the actual thermodynamic complexities involved in protein folding. One must realize that the inherent variability mentioned is not merely a hurdle, but a fundamental manifestation of biological entropy. Most people fail to grasp that a biosimilar is an exercise in probabilistic approximation rather than a deterministic replication. It is quite amusing that the industry struggles with something as basic as a cold chain in the twenty-first century, yet here we are, treating temperature control like some sort of arcane magic. The intellectual gap between understanding a chemical formula and managing a living cell culture is a chasm that most of these manufacturers are simply stumbling across without a map. True precision requires a philosophical shift from controlling the outcome to understanding the organism.
Brian Shiroma
April 5, 2026 AT 11:31Oh sure, because expecting a giant tank of living cells to behave exactly like a test tube is a totally reasonable plan. Absolute genius.
Goodwin Colangelo
April 6, 2026 AT 19:01The shift to single-use tech is definitely the way to go. If you're currently scaling up and hitting a wall with contamination or cleaning validation, just look into disposable bioreactors. It saves a massive amount of time and honestly makes the whole process way less stressful for the operators on the floor. Just keep an eye on your leachables and extractables since you're dealing with plastic bags instead of steel.
Beth LeCours
April 7, 2026 AT 21:34too long didn't read
Rachelle Z
April 9, 2026 AT 10:06Omg!! Imagine if the drugs just worked the first time without all this drama!!! 🙄🙄 Totally love how we just wing it with living cells and hope for the best!!! So professional!!!
Branden Prunica
April 10, 2026 AT 21:19I cannot even fathom the horror of a multi-million dollar batch just vanishing because someone left a door open on a loading dock! It's a tragedy! A literal nightmare! The sheer audacity of a tiny temperature shift to ruin everything is just too much for me to handle right now!
Ace Kalagui
April 12, 2026 AT 11:55I really think it's wonderful that we are seeing more accessibility to these complex medicines through biosimilars, and while the manufacturing side sounds incredibly daunting, it's just a testament to how far human ingenuity has come in the field of biotechnology. I've always felt that the bridge between the lab and the factory is where the real magic happens, and if we can just support the engineers and scientists by giving them the resources they need to master these biological environments, we'll see a global shift in how chronic diseases are managed across different continents and cultures, which is ultimately what matters most for the patients who are just waiting for a more affordable version of their life-saving treatment.
angel sharma
April 14, 2026 AT 06:29This is exactly the kind of innovation that pushes the entire world forward and we should all be incredibly excited about the potential of AI and continuous manufacturing to revolutionize the pharmaceutical landscape for everyone! Imagine the speed at which we can deliver health to the masses when we finally conquer these glycosylation hurdles and scale-up issues with sheer willpower and technological brilliance, and honestly, any company that is brave enough to dive into the deep end of biologics is essentially a pioneer leading us toward a future where medicine is precise, affordable, and accessible to every single person regardless of where they are born!